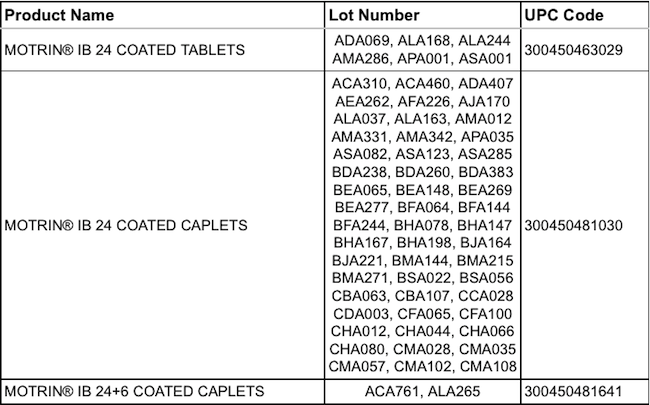
After more than a year of near-constant recalls, the folks at Johnson & Johnson’s McNeil Labs aren’t finished yet. This time, it’s a mere 12 million bottles of Motrin that are being recalled from store shelves because the pills may not dissolve as quickly as they are supposed to. [More]
mcneil laboratories

Worst Company In America Round One: CVS Vs. Johnson & Johnson
Here’s an interesting battle between a drugstore chain and the drug company whose products line the chain’s shelves — or at least they used to before they were all recalled. [More]

Latest Rolaids Recall Gives Johnson & Johnson Heartburn
Two weeks after recalling some cherry flavored Rolaids softchews for “uncharacteristic consistency or texture,” Johnson & Johnson’s beleaguered McNeil Laboratories division has issued a slightly larger Rolaids recall. And by “slightly,” we mean all lots of Rolaids Extra Strength Softchews, Extra Strength plus Gas Softchews, and Multi-Symptom plus Anti-Gas Softchews. [More]

The Johnson & Johnson Recall Wheel Lands On Mylanta
Because it has apparently already recalled every possible product made by its McNeil Labs division, Johnson & Johnson has moved over to its Merck products for its latest recall. This time, J&J has issued a recall on 12 Mylanta liquid products and one Alternagel product for trace amounts of alcohol that weren’t properly labeled on the packaging. [More]
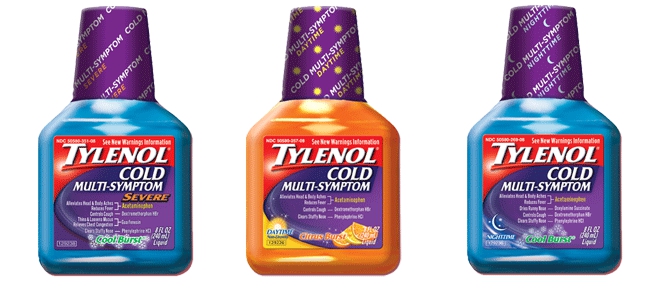
Thought They Couldn't Recall Any More Tylenol? You Were Wrong
In an apparent effort to make Tylenol products a rare commodity, Johnson & Johnson’s McNeil Laboratories division has issued a recall on 9 million bottles of its Tylenol Cold Multi-Symptom liquid beverages medicines for trace amounts of alcohol not mentioned on the label. [More]

Recalls Issued For Children's Benadryl, Motrin & Rolaids Softchews
Johnson & Johnson continues to earn its title as the recall king of the over-the-counter drug business with the announcement of three separate recalls totaling around 5 million units of Children’s Benadryl Allergy Fastmelt Tablets, Junior Strength Motrin Caplets, and Extra Strength Rolaids Softchews. [More]

Even More Tylenol Recalled Over Musty Odor
Johnson & Johnson’s 2010 continues to stink — literally — as the company announced yet another recall of its Tylenol painkiller over complaints of a musty/moldy odor. [More]