When it comes to over-the-counter pain pills, many people don’t even think to look at the expiration date on the side of the bottle. But a new class-action lawsuit claims that three of the biggest names in the (legal) drug business are deliberately putting early expiration dates on their products to encourage customers to throw them out and buy new ones. [More]
johnson & johnson

Lawsuit Accuses Big Pharma Of Using Bogus Expiration Dates To Trick Customers Into Buying More Meds

Johnson & Johnson Promises To Kick All Harsh Chemicals To The Curb By End Of 2015
Johnson & Johnson is going forward with plans to take out any of the nasty chemicals consumers have taken issue with recently, by pledging not only to remove “chemicals of concern” from baby products by 2013, but also to reformulate adult products with safer ingredients by the end of 2015. [More]

How I Took On Johnson & Johnson In Small Claims Court, And Won
Desitin, the diaper rash treatment, is not a nice-smelling substance. Joyce’s husband, Ben, could tell that much before using it to treat the aftereffects of some severe gastrointestinal distress. What he didn’t realize was that the unholy stench could not be defeated. It soaked through his clothing, and when he tried to launder those clothes, the smell didn’t go away: it spread to those other items as well. Annoyed at having to toss $600 worth of clothing, the couple contacted Johnson & Johnson, seeking compensation and encouraging the company to do more to deter people who do not currently wear disposable diapers from using the product. After some letters and an offer of $75 passed in the mail, they went to small claims court. And won. [More]

Court Orders Johnson & Johnson To Pay $1.2 Billion In Deceptive Marketing Case
Johnson & Johnson and a subsidiary have to pay for the way it marketed antipsychotic drug Risperdal, neglecting to mention its side effects while claiming it was safer and worked better than the competition. In Arkansas, one of 11 states in which J&J has faced legal battles over the drug, a judge ordered the company to pay $1.2 billion to make up for its alleged sins. [More]
Johnson & Johnson Ends 2011 Just At It Began — By Recalling More Stuff
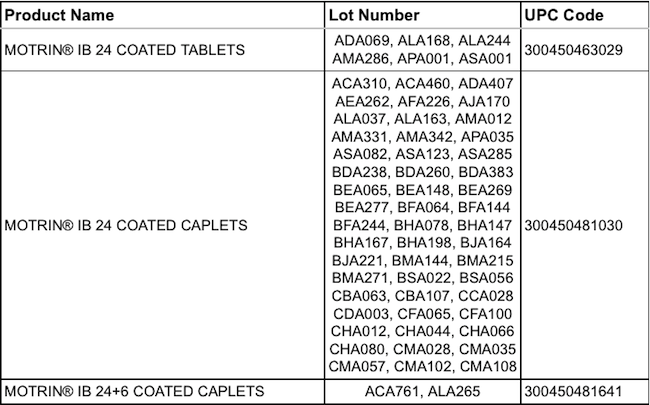
After more than a year of near-constant recalls, the folks at Johnson & Johnson’s McNeil Labs aren’t finished yet. This time, it’s a mere 12 million bottles of Motrin that are being recalled from store shelves because the pills may not dissolve as quickly as they are supposed to. [More]

O.B. Finally Apologizes To America For Tampon Shortage (With A Catch)
Last week, we told you the folks at o.b. tampons had issued an oddball, tongue-in-cheek apology in the form of a cheeseball ballad, complete with customizable video, for the many months that their products were off store shelves. Only problem was, that apology — and the coupon that came with it — was only available to Canadian shoppers. [More]

O.B. Tampons Apologizes To Canadian Customers… In Song Form
As many of you may remember from this time last year, o.b. tampons became one of many Johnson & Johnson products to disappear from store shelves. And for many, many months, store shelves remained o.b.-free, until they gradually began showing up in the spring. Now the tampon makers are saying “we’re sorry”… at least to its Canadian customers. [More]

Exxon Mobil Is The Most Profitable American Business By A Long Shot
This year’s Fortune 500 list is out, and while Walmart’s $421 billion in revenue may have beaten out Exxon Mobil’s paltry $354 billion, the oil giant beat out Big W where it matters most, profits. According to Fortune, the crude colossus made a whopping $30.4 billion in profits last year, nearly double what Walmart made and over $10.5 billion more than the next most profitable company on the list. [More]

Johnson & Johnson's Prescription Drugs Stink Too: 57,000 Bottles Of Topamax Recalled
If you thought Johnson & Johnson’s McNeil Labs division — the folks behind all of these recalled over-the-counter drugs — was the only branch of the company capable of creating stinky meds, you were wrong, as J&J has announced a recall of two lots of its prescription anitconvulsant Topamax for that now-classic “uncharacteristic odor.” [More]

Johnson & Johnson Settles With SEC & DOJ For $70 Million For Bribing Doctors Overseas, Paying Kickbacks To Iraq
Johnson & Johnson may have been eliminated from the Worst Company In America tournament, but the company’s craptastic year continues, as J&J has settled with the Securities and Exchange Commission and Dept. of Justice over allegations that it violated the Foreign Corrupt Practices Act by illegally bribing doctors in Europe and paying kickbacks to Iraq… At least it wasn’t another product recall. [More]

Worst Company In America Sweet 16: Johnson & Johnson Vs. WellPoint
In honor of April 1, we present this WCIA faceoff between a pair of fools — One that makes medicines no one can sell and the other that takes your insurance premium but offers you no insurance. [More]

Johnson & Johnson Keeps The Tylenol Recall Express Rolling
In advance of its Worst Company In America Sweet 16 bout against WellPoint, Johnson & Johnson has given us all a reminder of why they made the list to begin with, announcing yet another recall of a Tylenol product. [More]

Meet Your Worst Company In America Sweet 16!
After eight days and 16 first round battles, the WCIA steel cage is littered with the bones of those companies not crappy enough to continue on in the tournament. But the thrill of victory is fleeting for the remaining combatants, all of whom must square off again if they hope of crowning themselves the Worst Company In America! [More]

Worst Company In America Round One: CVS Vs. Johnson & Johnson
Here’s an interesting battle between a drugstore chain and the drug company whose products line the chain’s shelves — or at least they used to before they were all recalled. [More]
Government To Take Control Of Three Tylenol Plants
Following a slew of recalls that seemed to have pulled just about every Tylenol product from store shelves, three Johnson & Johnson plants responsible for the recalled goods are being put under the supervision of the FDA. [More]

Johnson & Johnson Recalls Even More Tylenol, Benadryl, Sudafed & Sinutab
The new year isn’t starting well for Johnson & Johnson. First, the OTC drug giant gets sued by the state of Oregon over allegations of a phantom Motrin recall. And now its McNeil Labs division has issued the latest in a lengthy list of actual recalls. This time, it’s for certain lots of Tylenol 8 Hour, Tylenol Arthritis Pain, Tylenol upper respiratory products, Benadryl, Sudafed PE, and Sinutab products distributed in the United States, the Caribbean, and Brazil. [More]

Latest Rolaids Recall Gives Johnson & Johnson Heartburn
Two weeks after recalling some cherry flavored Rolaids softchews for “uncharacteristic consistency or texture,” Johnson & Johnson’s beleaguered McNeil Laboratories division has issued a slightly larger Rolaids recall. And by “slightly,” we mean all lots of Rolaids Extra Strength Softchews, Extra Strength plus Gas Softchews, and Multi-Symptom plus Anti-Gas Softchews. [More]

The Johnson & Johnson Recall Wheel Lands On Mylanta
Because it has apparently already recalled every possible product made by its McNeil Labs division, Johnson & Johnson has moved over to its Merck products for its latest recall. This time, J&J has issued a recall on 12 Mylanta liquid products and one Alternagel product for trace amounts of alcohol that weren’t properly labeled on the packaging. [More]

