Johnson & Johnson Ends 2011 Just At It Began — By Recalling More Stuff
After more than a year of near-constant recalls, the folks at Johnson & Johnson’s McNeil Labs aren’t finished yet. This time, it’s a mere 12 million bottles of Motrin that are being recalled from store shelves because the pills may not dissolve as quickly as they are supposed to.
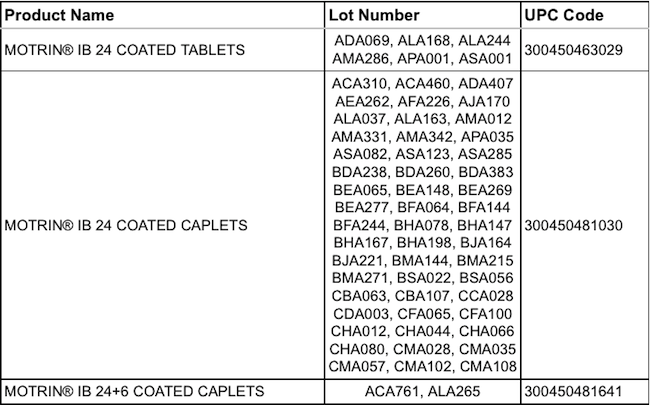
The recall involves certain lots (see below) of Motrin IB 24 count Coated Caplets, Motrin IB 24 count Coated Tablets and Motrin IB 24+6 count Coated Caplets.
J&J hastens to note that this is a retail-level recall, and not a consumer-level recall, indicating that this is not a safety issue. Instead, these pills are being pulled from retail shelves only.
Herewith the explanation for the recall:
McNeil is recalling these products because testing of product samples showed that some caplets may not dissolve as quickly as intended when nearing their expiration date. Out of an abundance of caution, we are recalling all the listed products since there is a chance they could experience a similar problem as they approach expiration.
Here are the recalled lots:
If you want to chat about this recall with someone at McNeil, give ’em a ring at 1-888-222-6036 (available Monday-Friday 8 a.m. to 8 p.m. Eastern Time).
For those feeling nostalgic, here’s a little Johnson & Johnson/McNeil recall round-up from the last year-plus:
Yet Another Tylenol Recall: 2.5 Million Cold Multi-Symptom Nighttime Rapid Release Gelcaps
Johnson & Johnson’s Prescription Drugs Stink Too: 57,000 Bottles Of Topamax Recalled
Johnson & Johnson Keeps The Tylenol Recall Express Rolling
Government To Take Control Of Three Tylenol Plants
Johnson & Johnson Recalls Even More Tylenol, Benadryl, Sudafed & Sinutab
Latest Rolaids Recall Gives Johnson & Johnson Heartburn
The Johnson & Johnson Recall Wheel Lands On Mylanta
Thought They Couldn’t Recall Any More Tylenol? You Were Wrong
Recalls Issued For Children’s Benadryl, Motrin & Rolaids Softchews
Want more consumer news? Visit our parent organization, Consumer Reports, for the latest on scams, recalls, and other consumer issues.

