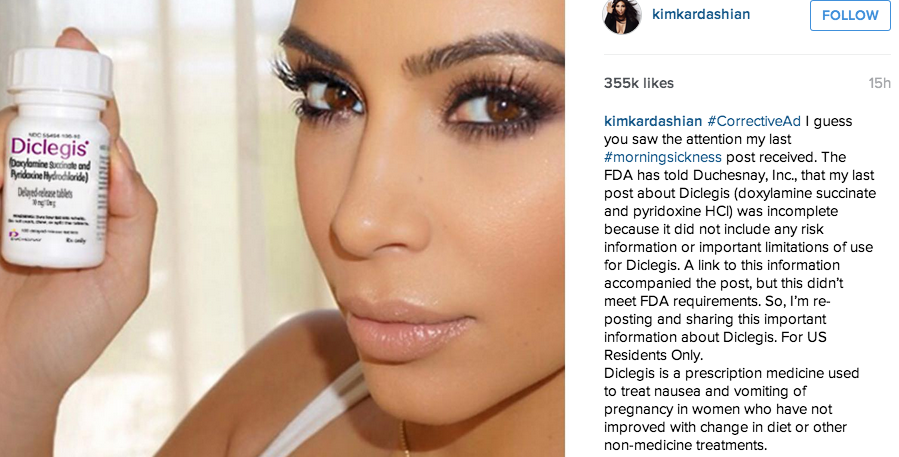
Weeks after federal regulators took issue with drug company Duchesnay for allowing mom-to-be Kim Kardashian to tout the benefits of its morning sickness pill on social media without properly disclosing the drug’s associated risk and limitations, the reality star posted updated endorsements, complete with acknowledgement of the pill’s side effects. [More]
Misbranding

FDA: Lack Of Info In Kim Kardashian’s Endorsement Of Morning Sickness Pill Puts Consumers At Risk
While celebrities get special treatment most places they go, there is no VIP pass that allows them to endorse a prescription drug without disclosing its associated risks and limitations. So when mom-to-be Kim Kardashian used social media to sing the praises of a morning sickness pill, it raised a red flag for federal regulators. [More]