Lawsuit Against CVS Claims Label On Advanced Eye Health Supplement Is Misleading
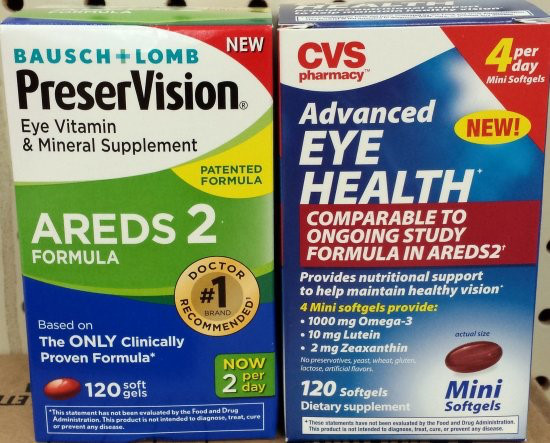
The Bausch + Lomb product on the left actually contains the eye health formula detailed in the NIH studies. The CVS product on the right mentions the formula, but lacks many of the essential components.
Age-related macular degeneration (AMD) is a leading cause of blindness in the U.S. As part of an effort to combat AMD, the National Institutes of Health have undertaken two studies — the Age-Related Eye Disease Studies (AREDS) — and have arrived at what the NIH believes is a specific formula of nutrients and vitamins that are helpful in slowing and reducing the risk of AMD.
And if you go into your drugstore, you’ll probably see some eye health supplements, like the Bausch + Lomb one above, that state they use the “AREDS 2” formula, referring to the most recent NIH study. Then you have products like the CVS brand Advanced Eye Health above that state they are ““Comparable to ongoing study formula in AREDS2.”
But the problem, according to the complaint [PDF] filed in a California court in Marin County on Tuesday, is that the CVS formula is “in no way comparable to the NIH study formula.”
The plaintiffs, two California men with AMD who purchased the CVS product, say that Advanced Eye Health lacks the vitamin C, vitamin E, zinc, and copper that the NIH studies deemed important to its formula.
Instead, the CVS product contains two plant-derived antioxidants — lutein and zeaxanthin — and omega-3 fatty acids.
However, in the conclusion for AREDS 2 [PDF], the NIH found that “Adding omega-3 fatty acids did not improve” on the original vitamins, beta-carotene, zinc and copper formula. And while the NIH concluded that lutein and zeaxanthin were safer than plain old beta-carotene, their addition to the formula “had no overall effect on AMD.”
The complaint alleges that since 2012, “CVS has engaged in a widespread marketing campaign to deceive… reasonable consumers about the nature, composition, and nutritional and health benefits of its Advanced Eye Health supplement in order to make the product more desirable to those consumers, increase sales, and gain market share.”
The plaintiffs say that “Without a degree in nutrition science and the AREDS 2 study on hand, no consumer could discover CVS’s deception.”
The lawsuit seeks class-action status for anyone who purchased CVS’s Advanced Eye Health supplement in the last four years.
The plaintiffs are represented by Amanda Howell, litigator for the Center for Science in the Public Interest.
“CVS is blatantly cheating older consumers who may fear developing age-related macular degeneration,” said David Schardt, senior nutritionist at CSPI in a statement.
UPDATE: CVS has provided Consumerist with the following statement —
“As this matter involves pending litigation, we cannot comment on the allegations in the complaint. However, when this product was launched in July 2012, the Age-Related Eye Disease Study 2 (AREDS2) was ongoing. CVS/pharmacy Advanced Eye Health dietary supplement was formulated with the same nutrients that were the primary focus of the study. CVS/pharmacy removed this statement from the product once the results of the AREDS2 study were released.”
Want more consumer news? Visit our parent organization, Consumer Reports, for the latest on scams, recalls, and other consumer issues.

