The new year isn’t starting well for Johnson & Johnson. First, the OTC drug giant gets sued by the state of Oregon over allegations of a phantom Motrin recall. And now its McNeil Labs division has issued the latest in a lengthy list of actual recalls. This time, it’s for certain lots of Tylenol 8 Hour, Tylenol Arthritis Pain, Tylenol upper respiratory products, Benadryl, Sudafed PE, and Sinutab products distributed in the United States, the Caribbean, and Brazil. [More]
sudafed
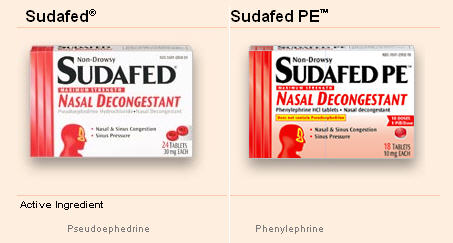
Sudafed PE: Pregnant Women Should Watch Out For Reformulated Medicines
Not to malign the good name of Sudafed, but it seems there is some debate out there as to whether or not pregnant women should take the reformulated “Sudafed PE.” Obviously, we are in no way doctors, nor do we know the first damn thing about pregnant woman, but we do know that several medicines have reformulated in order to stay on the shelves and we wanted to make sure people knew about it. Because Sudafed contains an ingredient used to make meth, some states now require it to be stored behind the pharmacist’s counter. Reasonable enough. The confusion comes in when people don’t realize that the new “on-the-shelf” Sudafed contains different ingredients than the old Sudafed.


