Poor Novartis: things just keep going wrong at their plants. Last year, they had multiple recalls, including many of their over-the-counter drugs and a line of birth control pills. Now, the company has recalled liquid Triaminic and TheraFlu products because the products’ caps aren’t childproof. The Consumer Products Safety Commission has heard about twelve kids who have opened the bottle themselves, four of whom had a taste. One needed medical attention. [More]
otc

Where Has My Slow Fe Iron Supplement Gone?
Cheryl takes iron supplements. She has iron deficiency anemia, and the vast majority of iron supplements on the market make her ill. She’s come to rely on Slow-Fe, made by Novartis, to keep her iron levels up and her digestive system functioning. Then Slow-Fe disappeared. Her regular pharmacist can’t find any to order, and the only sources online are re-sellers with expired products. Remembering our past coverage of catastrophic OB tampon, Eggo waffle, and Morningstar veggie dog shortages, Cheryl wrote to us, asking for help. Could we help her figure out where her precious iron had gone? [More]

Johnson & Johnson Hired Fake Shoppers To Buy Up Bad Motrin, Avoid Public Recall
Ever since the FDA and Congress started asking Johnson & Johnson to explain why it keeps recalling medicine, there have been references to an unpublicized “recall” that happened in November 2008. Last month, at a hearing of the House Oversight and Government Reform Committee, a J&J executive swore that the company didn’t mean to mislead anyone. It turns out that wasn’t exactly accurate: Bloomberg has obtained emails from J&J’s company, McNeil Consumer Healthcare, that show executives knew the secret recall would trigger an FDA reaction if the agency got wind of its full scope. [More]

Save Money On Head Lice Removal
You’re a good Consumerist. You make your own kids at home. You grow your own lice in a coffee can you found on the street. You dump the lice on the kids’ heads before you send them off to school. After all that, the last thing you want to do is spend a fortune on lice removal treatments, right? You’re in luck: the New York Times says you don’t have to spend a lot of money de-lousing your itchy little child. [More]
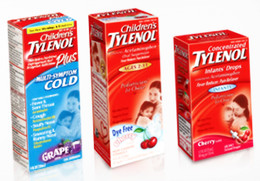
Tylenol Recall Factory Was Staffed With Undertrained Temps
The manufacturing plant that has been the cause of Johnson & Johnson’s latest in a string of recalls has already been described as dirty and poorly maintained. It turns out that it was also staffed with temps and contract employees who weren’t properly trained, according to tax records and an FDA inspection report filed earlier this year. [More]

FDA: McNeil Plant That Made Recalled Tylenol Is A Dirty Stinkpot With No Quality Control
One of the implied promises of a brand name, especially when it comes to drugs, is you can expect higher quality, but maybe that doesn’t apply when it comes to McNeil products.The FDA says the plant that produced the recently recalled children’s Tylenol, Motrin, Zyrtec and Benadryl, was using raw materials that were contaminated with bacteria. The plant also lacked adequate quality-control procedures and was dirty. So far none of the recalled medicine has tested positive for bacterial contamination, but the FDA report suggests that the contaminated material was used to make the recalled lots. The plant has been shut down indefinitely. [More]
FBI Reopens Infamous "Tylenol Murders" Case
If you’ve ever wondered why medicines have tamper-proof seals — there’s one reason: an group of still unsolved murders over a quarter of a century old. In September of 1982, cyanide-laced Tylenol killed seven people in the Chicagoland area. Despite a nationwide recall and investigation, no one was ever charged with the crime. Now the FBI has reopened the case.

FDA Considers Banning OTC Cough Medicines For All Children Under 6
Pediatricians are asking the FDA to recall all OTC cough medicines for children under six years old, and the FDA is holding a public hearing on the subject today. One reason this has only recently become an issue is that when the FDA originally set rules for OTC cough medicines, they were based only on studies for adults, not kids, writes the Associated Press. Although there’s a low risk of unintentional overdose—the AP says about 7,000 children are admitted to ERs each year—the other issue is that there’s very little evidence that they’re effective.

Target's Generic Meds Are Maybe Too Generic
We’re big fans of Target’s smart approach to package design for medicine. They may want to give a little more thought to their OTC generics, however—how about using more distinct labeling for the children’s line, for example? One reader explains why this would be a lot safer.

Zyrtec Goes Behind-The-Counter
Zyrtec users can now buy it without a prescription—but they’ll have to show ID because it going to be sold from behind the counter with the other meth supplies. [Associated Press]

Safety Experts Urge FDA To Ban OTC Cough And Cold Meds For Kids Under 6
The New York Times is reporting that safety experts are urging the FDA to consider a ban of all “over-the-counter, multisymptom cough and cold medicines for children under 6.”

FDA Says No Cough Syrup For Toddlers Without Doctor Approval
“Questions have been raised about the safety of these products and whether the benefits justify any potential risks from the use of these products in children, especially in children under 2 years of age,” the agency said.



