Looks like o.b. brand tampons, missing from shelves for months because of an unspecified Johnson & Johnson “supply chain interruption,” are slowly making their way back into stores. The company announced back in February that they were back, and a reader saw them at their Fred Meyer in Arkansas, but by mid-march another reader said they hadn’t returned. Here it’s April and o.b.’s haven’t returned to my go-to supermarket here in Brooklyn but then I spotted these at a local upmarket pharmacy this weekend. At this point it’s a question of what suppliers your store is working with. Ask your store manager if you still don’t see them yet. [More]
johnson and johnson

O.B. Tampons Are Back
Johnson and Johnson say new shipments of o.b. tampons have started going out and they should start showing up in a few days or weeks, depending on your store’s restock schedule. You can also get them online at Amazon.com, drugstore.com and soap.com. o.b. Ultras will not be returning as they’ve been discontinued. [More]
Oregon Sues J&J For Secretly Recalling Motrin
Instead of issuing a recall, after Johnson and Johnson discovered its Motrin caplets were defective, they hired a contractor to go around the country buy up all the drugs. It was a “secret recall” that left the bad medicine on shelves for months for consumers to buy. Now the Oregon AG is suing J&J. The story broke when one of the guys hired to do the buying faxed regulators the instructions J&J gave him. [More]

Where Did All The OB Tampons Go?
If you’re a fan of OB Tampons, better start your hoarding. They have been taken off the shelves and won’t return until 2011, due to an unspecified “manufacturing update.” UPDATE: O.B. Tampons Are Back [More]
Thought They Couldn't Recall Any More Tylenol? You Were Wrong
In an apparent effort to make Tylenol products a rare commodity, Johnson & Johnson’s McNeil Laboratories division has issued a recall on 9 million bottles of its Tylenol Cold Multi-Symptom liquid beverages medicines for trace amounts of alcohol not mentioned on the label. [More]

Recalls Issued For Children's Benadryl, Motrin & Rolaids Softchews
Johnson & Johnson continues to earn its title as the recall king of the over-the-counter drug business with the announcement of three separate recalls totaling around 5 million units of Children’s Benadryl Allergy Fastmelt Tablets, Junior Strength Motrin Caplets, and Extra Strength Rolaids Softchews. [More]

FDA: Hey America, Stop Overdosing On Painkillers!
Look, we know this recession is tough and all, but you’ve gotta lay off the NyQuil and Theraflu or the FDA will stuff them behind a counter, ok? Seriously, an advisory panel is meeting today, and already voted to reduce the maximum daily dose of Tylenol and other painkillers. They might even slap scary “black box” warnings on all over-the-counter painkillers to dissuade you acetaminophen addicts from overdosing.

Baby Products: Now With Formaldehyde!
More than half of the baby products recently tested by the Campaign for Safe Cosmetics were found to contain trace levels of formaldehyde and dioxane. Though the study didn’t accuse Johnson & Johnson of dumping barrels of the potential carcinogens directly into their baby products, the dangerous chemicals can form during the manufacturing process as other ingredients break down. The full list of 48 tested baby shampoos, lotions, soaps, and wipes—including some well-known products you probably have on your shelf—inside.
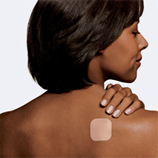
J&J Has Paid Over $68.7 Million In Ortho Evra Lawsuits
Johnson & Johnson has paid out over $68.7 million in private settlements in Ortho Evra birth-control related claims. The Public Citizen’s Health Research Group is petitioning the FDA to take it off the market. The most common complaints among users of the contraceptive patch that adheres to the skin are blood clots in the legs, blood clots in the lungs, and hearts attacks/strokes. But hey, at least you don’t have a baby. [Bloomberg]

14 Children's Cold Meds Withdrawn From the Market
Johnson & Johnson today recalled several infant cough and cold medicines, citing “rare instances of misuse leading to overdose, particularly in infants under two years of age.”



