When you buy a dietary supplement, you never know quite what you’re getting, because supplement manufacturers don’t have to prove to the Food and Drug Administration that their products work — or are even safe — before putting them on the market. GNC, the world’s largest dietary supplement retailer, has now agreed to try to ramp up efforts to ensure that the products it sells are safe and legal. [More]
GNC

GNC Looking To Sell Itself, Other Restructuring Options
Amid declining sales and increased scrutiny on the supplements industry, GNC Holdings is looking to either restructure its business or sell itself.
[More]
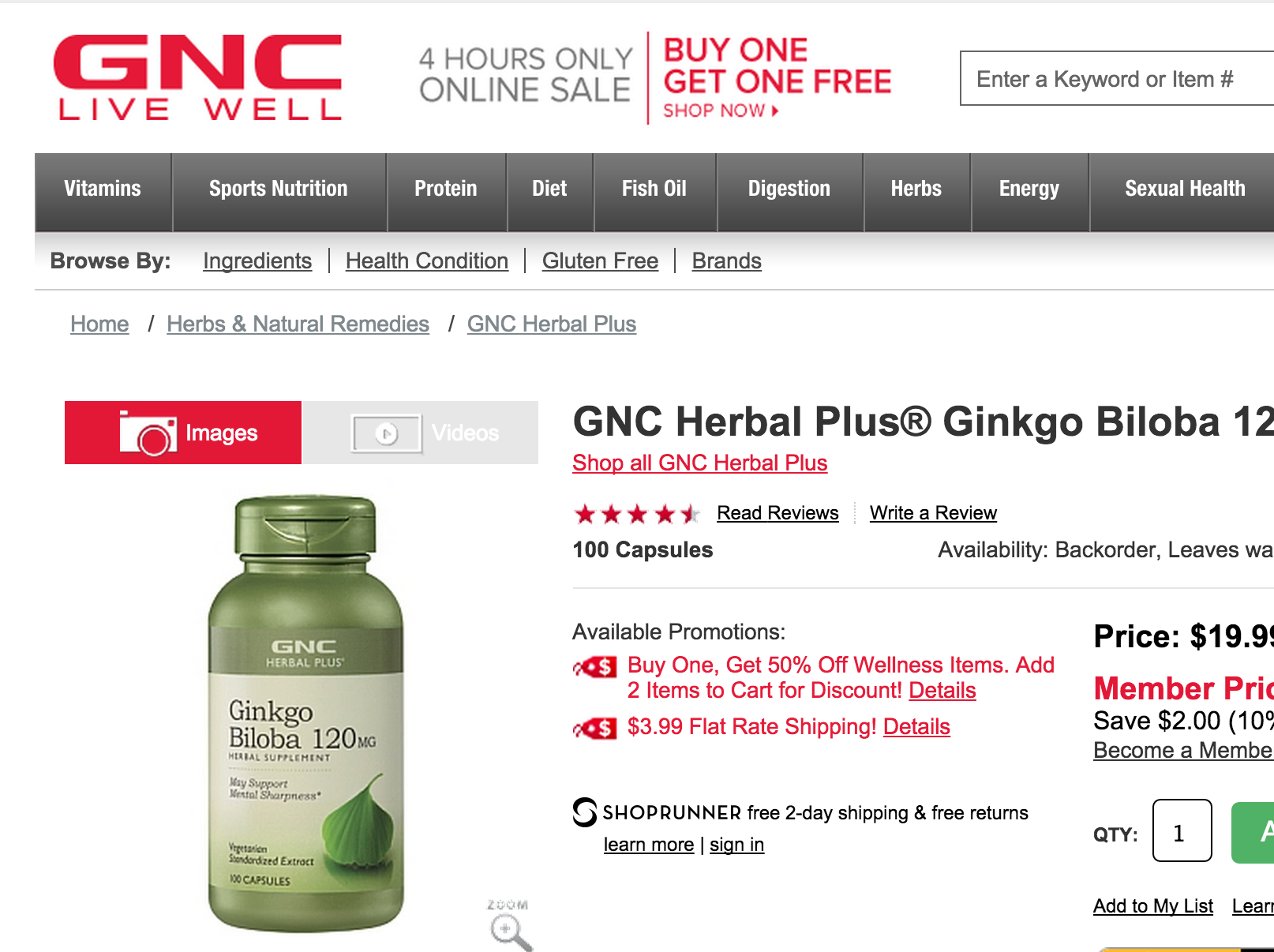
GNC Agrees To DNA, Allergen Testing Of Herbal Supplements
Earlier this year, New York state Attorney General Eric Schneiderman sent cease-and-desist letters to several major retailers, including Target, Walgreens, and GNC, after genetic testing of store-brand herbal supplements found that many of these products did not contain the herbal ingredients they purported to include, and that some contained potentially dangerous allergens. Today, GNC and Schneiderman announced an agreement that will apply stricter testing standards — more rigorous than those required by the federal government — to the company’s supplements. [More]

NY Asks Stores To Halt Herbal Supplements After Tests Show Advertised Herbs Not Present
When you buy an herbal supplement that says “echinacea” or “ginko boloba” on the label, you may expect that it contains some additional ingredients beyond the advertised herbs, but you should be confident that those herbs are present. However, DNA tests commissioned by the New York state Attorney General found evidence that many herbal products may not contain what they advertise. [More]

Beware Online Discounts That Vanish As You Check Out
Omie Ismail at LiveCheap warns online shoppers to look out for discounts that disappear before you finalize purchases. [More]


