Here’s Why The FDA’s Antibiotics-In-Agriculture Policy Is A Charade Image courtesy of Great Beyond
Let’s take a moment to compare the drug industry’s non-reaction to the FDA guidance on antibiotics to the TV industry’s overheated response to streaming video startup Aereo.
The major TV networks, which give their product away for free to anyone with an antenna, spent huge amounts of money filing lawsuits and getting their case before the Supreme Court, all in an effort to stop a startup with a small customer base and service in a limited number of markets.
The networks acted immediately and disproportionately to an incredibly minor threat because in the long run it could have set a precedent that may have resulted in a larger loss to the broadcasters. Just the notion of an Aereo victory led top network execs to speak publicly about actually pulling their channels from the airwaves and going cable-only. This sort of all-out blitz is what most of us have come to expect from industries that sniff any possible harm in the form of competition or regulation.
Meanwhile, antibiotics for farm animals account for around 80% of all antibiotics sold in the U.S. each year. And most of these drugs are purchased solely for the purpose of promoting tissue growth in the animals; they also have the effect of sometimes allowing animals to exist in filthy, cramped conditions (at least until the drugs are rendered ineffective by antibiotic-resistant microbes).
But when the FDA announced last fall that it was finally doing something about the overuse of these drugs, the pharmaceuticals industry didn’t run to the Supreme Court; it didn’t drop a few pills in a politician’s pocket to introduce new legislation to override the guidance. In fact, the pharma pholks not only agreed to the guidance, they said it was no big deal.
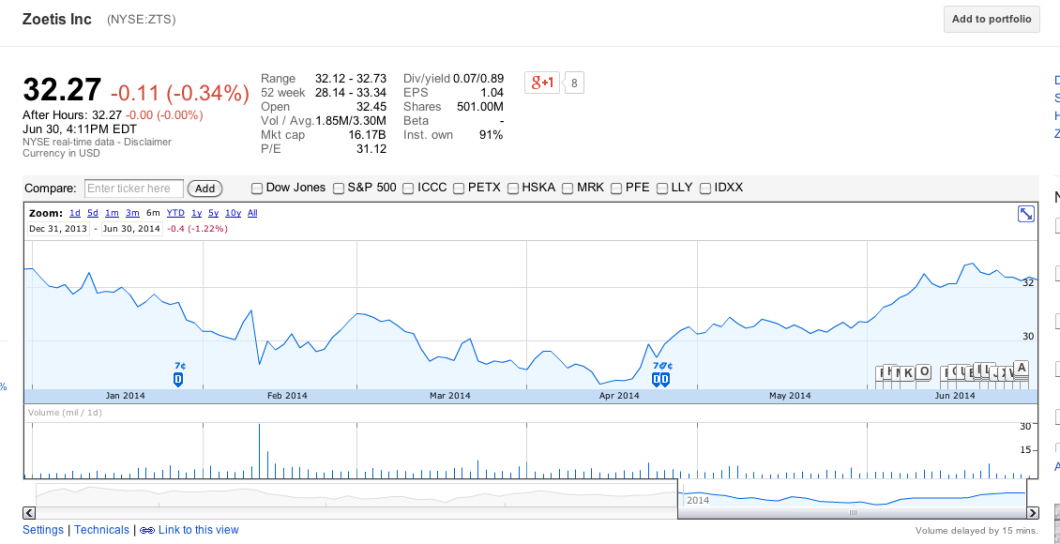
The CEO of Zoetis actually said at the time that the FDA guidelines — which are completely voluntary — “will not have a significant impact on our revenues.”
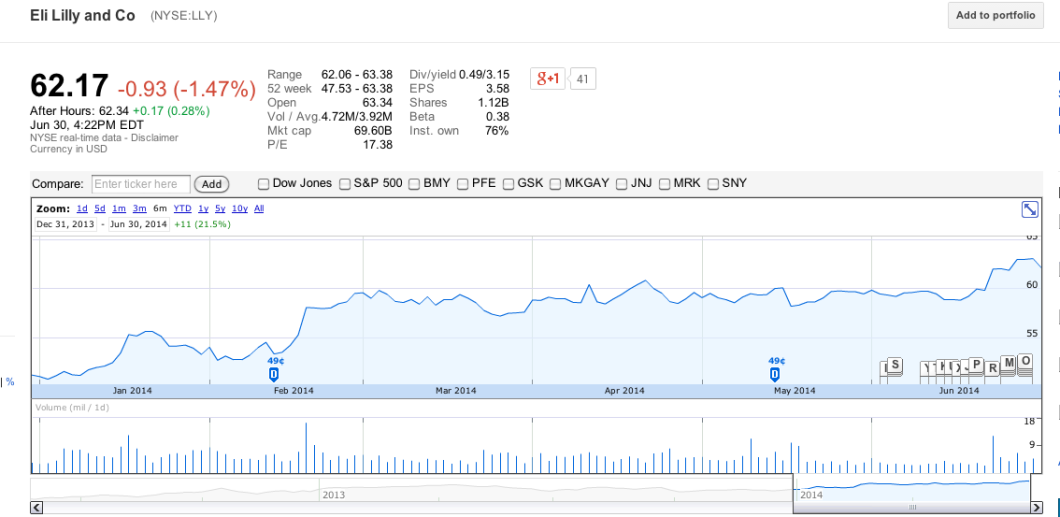
As you can see below, Zoetis’s stock price is virtually the same as it was six months ago, and has actually improved since April, implying that investors aren’t freaking out about the company losing a lot of business from the new FDA guidelines:
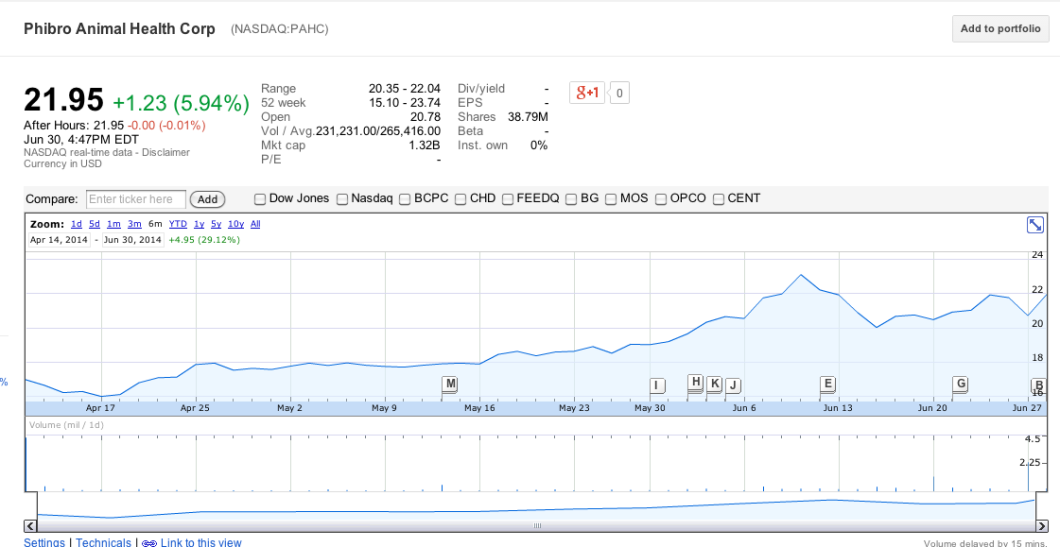
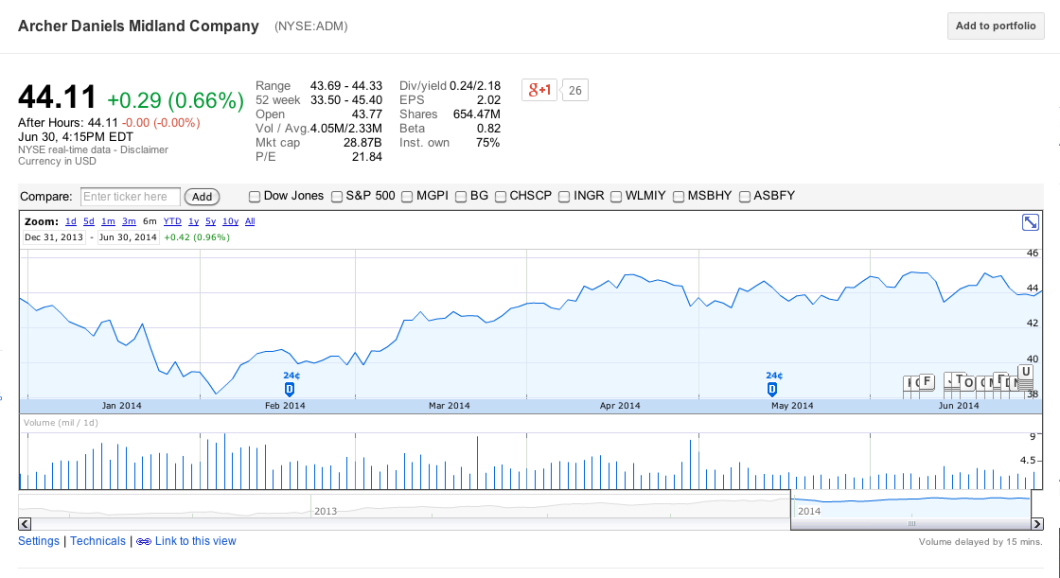
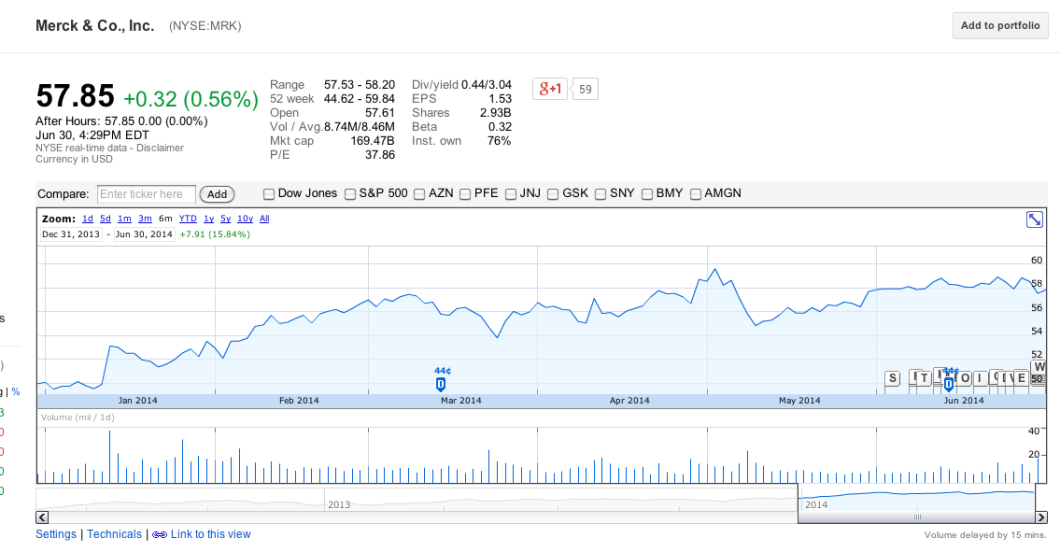
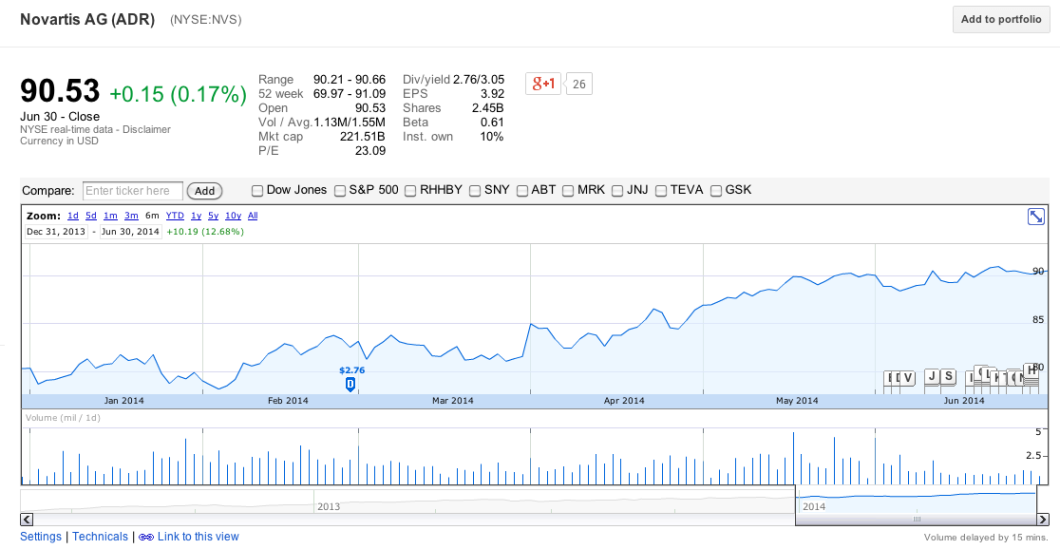
And while most of the 26 companies involved in this guidance are privately held, none of the public companies — like ADM, PAHC, Merck, Eli Lilly, Novartis — impacted by the FDA policy appear to be worse for wear:
Granted, most of these businesses have many other sources in addition to antibiotics, and many of them do business outside of the U.S., but the fact that not one of them has taken a hit to their stock prices indicates to us that investors are not concerned about the longterm effect that the FDA guidance will have.
So it’s with a huge grain of salt that we take today’s happy report from the FDA that every company that it believes to be impacted by the guidance has “agreed to fully engage” in the FDA phase-out of medically unnecessary drugs and phasing in of veterinary oversight.
But again, one has to wonder why drug companies that would ostensibly be harmed by stricter regulation be playing along so complacently.
“If something sounds too good to be true – it usually is,” says Congresswoman Louise Slaughter of New York, who has been a very vocal critic of the FDA guidance. “It would take an extraordinary leap of faith to believe that asking pharmaceutical companies to change the labeling on packages of antibiotics will result in a tangible reduction of antibiotic overuse on the farm.”
The Congresswoman points out that the guidance has no sort of reporting requirement, nor does it set any quantitative limits on the use of these drugs; it just requires farmers to change the reason they use for buying the antibiotics.
“Until the FDA comes up with an enforceable, measurable guidance that will actually limit the amount of antibiotics used in agriculture production, we will continue to erode the effectiveness of antibiotics until we reach a point where routine infections like strep throat become fatal and life-saving surgeries that require antibiotics to stave off infection become obsolete,” explains Slaughter.
Want more consumer news? Visit our parent organization, Consumer Reports, for the latest on scams, recalls, and other consumer issues.