Reader C. was cruising by the beer cooler at his local Hannaford supermarket when he noticed an interesting cross-promotion. There was a mini-shelf attached to the outside of the refrigerator case door, where you might expect to see maybe bottles of chocolate sauce near the ice cream. This mini-shelf in the beer case had… acetaminophen? That’s a pain reliever that can cause liver damage in not-so-high doses, which you shouldn’t combine with alcohol. [More]
acetaminophen
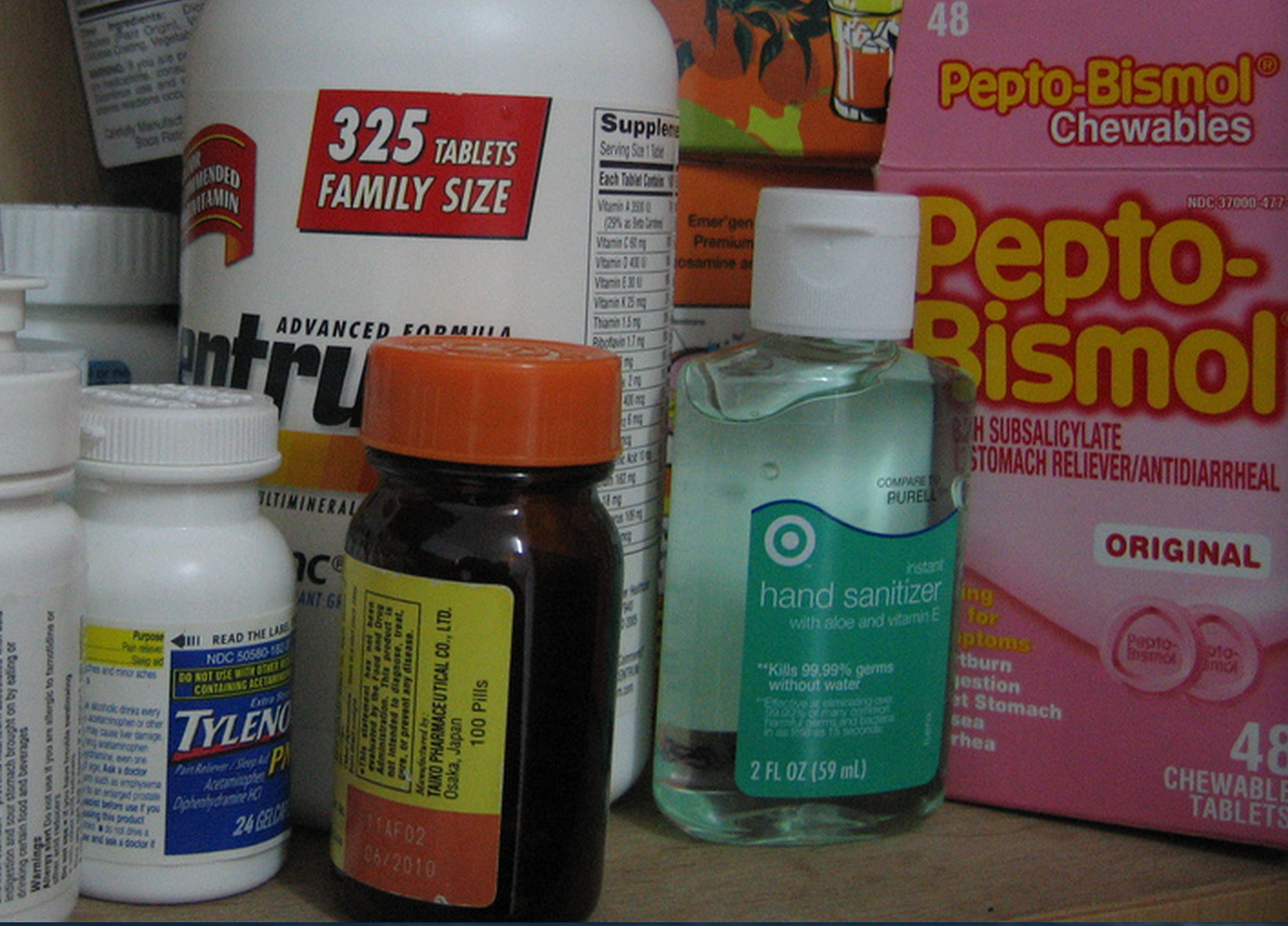
We Know How To Read Over-The-Counter Drug Labels, But We Don’t Always Understand The Potential Dangers
When you buy some Tylenol or NyQuil, you probably know — or find out easily from looking at the label — that these products contain acetaminophen. But knowing the names of active ingredients in an over-the-counter drug isn’t the same as knowing how those ingredients will interact with other drugs or what could happen if you take too much. A new study looks at the apparent disconnect between reading a label and understanding the potentially dangerous implications of what you’re taking. [More]

What’s The Difference Between All The Many NyQuil Variations?
For decades, sick people in search of a night’s rest — and high school kids in search of something to amuse themselves with — took Vicks NyQuil, and eventually woke up, often feeling like they’d hibernated for a season. Then they introduced DayQuil, which takes away all the fun of NyQuil, but supposedly lets you do your job without nodding out mid-meeting. More recently, Vicks added ZzzQuil and the bizarrely named QlearQuil, but what the H-E-double-hockey-sticks are they all about? [More]

FDA Warns Doctors, Pharmacists Against Prescribing High Doses Of Acetaminophen
Even though many people pop Tylenols or slug NyQuil without thinking about the consequences, too much acetaminophen can wreak havoc on one’s liver. Today, the Food and Drug Administration has asked health care professionals to please stop prescribing and dispensing dosages of medicine that contain more than 325 mg of acetaminophen, saying the risk to a patient’s liver is higher than any additional benefit of the drug. [More]

Valve on Kids’ Medicine Bottles Could Prevent Overdose Deaths, But Costs Money To Install So Never Mind
Acetaminophen, best known under the brand name Tylenol, is an incredibly common medication for children. While it’s safe at the recommended dose, it can be fatally harmful when taken in excess and the overdose threshold is surprisingly low. [More]

13 Medications You May Not Realize Contain Acetaminophen
For those who haven’t been paying attention — either in life generally or to new reporting on Tylenol regarding the potential dangers of acetaminophen — it’s time to snap to. Because I’m sure I’m not the only one reading the new report about children dying from Tylenol overdoses to feel like I’ve been a bit out of the loop on acetaminophen. And in this scenario, confusion can lead to accidental overdoses. [More]

Children Died Because Johnson & Johnson Insisted On Selling Two Types Of Kids’ Tylenol
At the intersection of bad marketing, inept regulation, and unwitting consumers, you’ll find the graves of young children, just some of the infants who, according to a new report from ProPublica, have become ill over the decades because Johnson & Johnson and other makers of acetaminophen-based painkillers insisted on selling two youth-targeted varieties of the drug while the FDA did what it does best — nothing. [More]
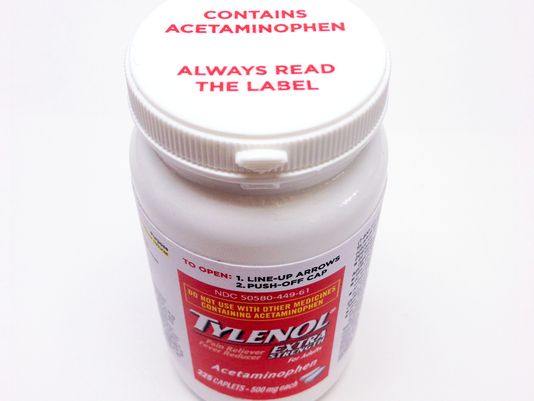
Johnson & Johnson Reveals New Acetaminophen Warning Labels On Bottles Of Tylenol
Johnson & Johnson has revealed a new bottle cap design for Tylenol products sold in the U.S. which will warn consumers in bright red lettering that the medication contains acetaminophen. The labels will alert users to the potentially fatal risks of taking too much Tylenol, which is an unusual step for a company to make. [More]
FDA Links Acetaminophen To Nasty, Possibly Fatal Skin Reactions (But Try Not To Freak Out)
Because no weekend can get started without news of a possible scary drug interaction, the FDA has issued a consumer alert warning that, in extremely rare cases, use of the popular over-the-counter painkiller acetaminophen (used in Tylenol and many others) can cause “rare but serious skin reactions,” including three serious skin diseases with symptoms like rash, blisters and widespread damage to the surface of skin. Yuck. [More]

Study: Painkiller Could Be Liver Killer
Over-the-counter painkillers seem harmless enough, but like any drug, they can cause plenty of pain of their own if overused. According to a study, popping too much acetaminophen over an extended period of time could cause liver failure and possibly lead to death. [More]
FDA Asks Prescription Drug Companies To Limit Amount Of Acetaminophen
Worried about the possibility of liver damage from over use, the U.S. Food and Drug Administration has asked manufacturers of prescription drugs containing acetaminophen to limit the amount of acetaminophen to no more than 325 mg in each tablet or capsule. [More]

Girl "Grows Back Face" After Pain Relief Pill Made It Turn Black And Fall Off
19-year old Eva Uhlin has regrown her face, chest, arms, back and stomach. A rare reaction to a couple pills of pain reliever acetaminophen, the main ingredient in Tylenol, caused her to develop Toxic Epidermal Necrolysis, and large parts of her skin turned black and fell off. [More]
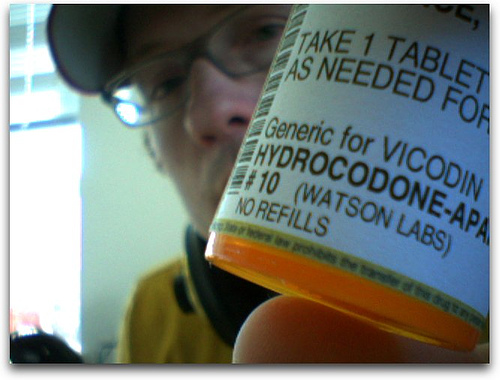
FDA May Take Vicodin And Percocet Off The Market
Bad news for Dr. Greg House and other, non-fictional chronic pain patients. The FDA advisory panel that met yesterday about the effects of excessive doses of acetaminophen made another recommendation to the FDA—to take popular painkillers Vicodin and Percocet (and their generic versions) off the market because of the effect both drugs can have on the liver when taken for extended periods. The FDA will most likely follow this recommendation.
FDA Calls For Stricter Warnings on Tylenol and Other OTC Drugs
“Non-prescription pain relievers used by millions of U.S. consumers need stronger health warnings regarding liver or stomach risk, the Food and Drug Administration said Tuesday.” The drugs in questions are acetaminophen (Tylenol), and NSAIDs like ibuprofen (Advil, Motrin). In particular acetaminophen is associated with liver problems.


