After FDA Warning, Kim Kardashian Posts Corrected Endorsement Of Morning Sickness Pill
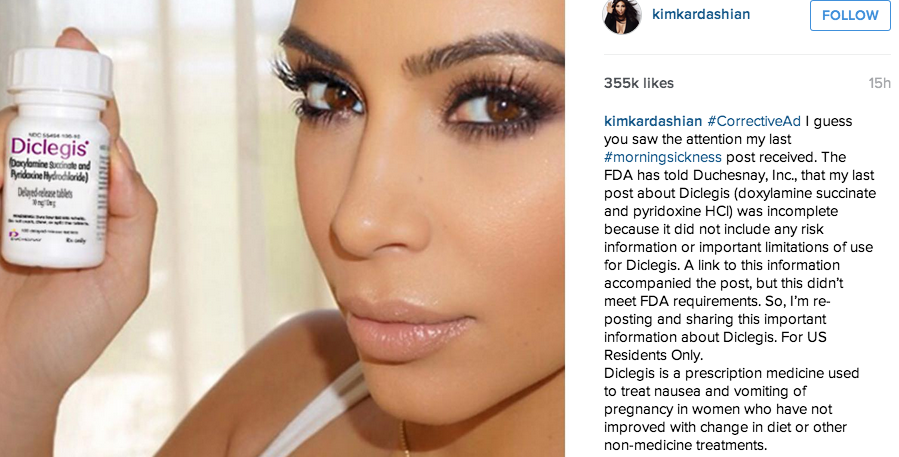
Kim Kardashian issued a corrected endorsement for morning sickness pill after the FDA took issue with a first post’s lack of acknowledgement of associated safety risks.
The new “#CorrectiveAd” posts – which were quickly pushed down the page thanks to Kardashian’s posts about the MTV Video Music Awards – were mandated by the FDA after the agency issued a warning letter to Duchesnay in early August saying the lack of safety information included in Kardashian’s original July posts put consumers at risk.
“I guess you saw the attention my last #morningsickness post received,” an Instagram post from the celebrity states. “The FDA has told Duchesnay, Inc., that my last post about Diclegis (doxylamine succinate and pyridoxine HCl) was incomplete because it did not include any risk information or important limitations of use for Diclegis. A link to this information accompanied the post, but this didn’t meet FDA requirements. So, I’m re-posting and sharing this important information about Diclegis.”
According to the FDA letter, a review of the first endorsements by Kardashian found the posts to be “false or misleading in that it presents efficacy claims for Diclegis, but fails to communicate any risk information associated with its use and it omits material facts.”
As a result, the FDA claims the post misbrands the pill, which makes its distribution a violation of the Federal Food, Drug, and Cosmetic Act.
Regulators told Duchesnay that the misleading messages – which were quickly deleted by Kardashian in August – be corrected in the same format they were originally posted – meaning on Instagram, Twitter and Facebook.
While the first posts included a link to the company’s website and Duchesnay sent out a press release providing more details about the partnership including information on Diclegis, the FDA claimed those steps weren’t enough to mitigate misinformation.
Kardashian addressed those issue in her corrected ads on Sunday. The rather lengthy post goes on to include limitations – “Diclegis has not been studied in women with hyperemesis gravidarum” – and important safety information including a number of other medications that shouldn’t be taken in conjunction with the pill.
Here’s the full text of the corrected ad:
#CorrectiveAd I guess you saw the attention my last #morningsickness post received. The FDA has told Duchesnay, Inc., that my last post about Diclegis (doxylamine succinate and pyridoxine HCl) was incomplete because it did not include any risk information or important limitations of use for Diclegis. A link to this information accompanied the post, but this didn’t meet FDA requirements. So, I’m re-posting and sharing this important information about Diclegis. For US Residents Only.
Diclegis is a prescription medicine used to treat nausea and vomiting of pregnancy in women who have not improved with change in diet or other non-medicine treatments.
Limitation of Use: Diclegis has not been studied in women with hyperemesis gravidarum.Important Safety Information
Do not take Diclegis if you are allergic to doxylamine succinate, other ethanolamine derivative antihistamines, pyridoxine hydrochloride or any of the ingredients in Diclegis. You should also not take Diclegis in combination with medicines called monoamine oxidase inhibitors (MAOIs), as these medicines can intensify and prolong the adverse CNS effects of Diclegis.The most common side effect of Diclegis is drowsiness. Do not drive, operate heavy machinery, or other activities that need your full attention unless your healthcare provider says that you may do so. Do not drink alcohol, or take other central nervous system depressants such as cough and cold medicines, certain pain medicines, and medicines that help you sleep while you take Diclegis. Severe drowsiness can happen or become worse causing falls or accidents.
Tell your healthcare provider about all of your medical conditions, including if you are breastfeeding or plan to breastfeed. Diclegis can pass into your breast milk and may harm your baby. You should not breastfeed while using Diclegis.
Additional safety information can be found at http://www.DiclegisImportantSafetyinfo.com or http://www.Diclegis.com. Duchesnay USA encourages you to report negative side effects of prescription drugs to the FDA. Visit http://www.fda.gov/medwatch or call 1-800-FDA-1088.”
Want more consumer news? Visit our parent organization, Consumer Reports, for the latest on scams, recalls, and other consumer issues.

